LATEST UPDATES
Sitero Unveils the Latest Version of Mentor RTSM!
Enhanced self-service capabilities and advanced reporting.
Designed to empower clinical research teams, this cutting-edge technology streamlines trial management with greater efficiency, control, and real-time insights — ensuring seamless execution at every stage.
Mentor RTSM Key Metrics
100+ CLIENTS | 300+ TRIALS | 10,000+ SITES | 50+ COUNTRIES SERVED

Why Mentor RTSM?
Configurable for complex protocols
Built for mid-study amendments
Connected to your trial stack
SPEED
FASTER
RTSM study startup
(median reduction in build + UAT time across PhII studies)
COST
LOWER
RTSM admin costs
(When compared to leading RTSM vendors)
TIME
DECREASE
in time spent on supply management
QUALITY
DECREASE
in wasted IP
(Average reduction in expired/wasted IP in supply constrained studies)
DATA
FEWER
IP stockouts at sites
(Across global, multi-site programs)
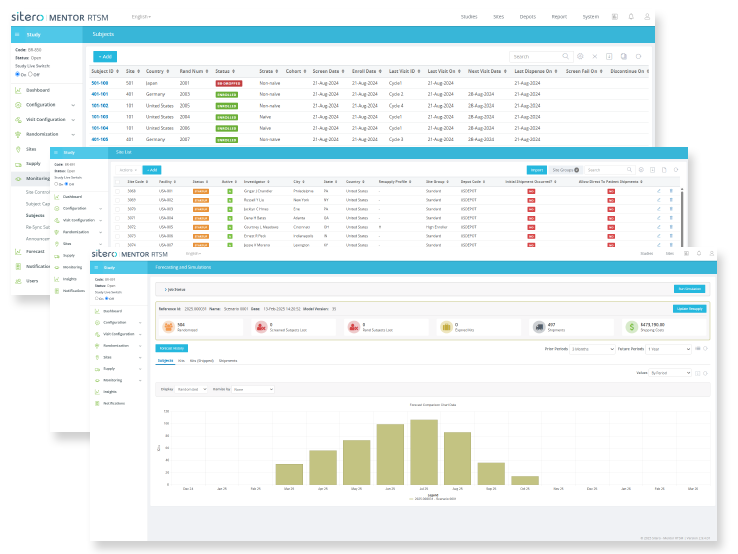
Mentor RTSM Features
Mentor RTSM delivers unmatched speed, flexibility, and control with real-time inventory tracking, predictive resupply, and full support for complex study designs.
Agile No/Low Code Implementation
Out of the Box Support for Complex Protocol Designs
No/Low Code
Mid-Study Changes
Out of the Box Integration with EDC and eConsent
Transparent Realtime Actionable Reporting
Key Differentiators
Smart automation, advanced logistics, and total accountability—helping sponsors stay in control across global studies.
Testimonial
“Transitioning to the updated RTSM platform has been one of the smoothest tech rollouts we’ve experienced. Sitero worked closely with our team to ensure every study build was aligned with our internal processes—and the predictive supply tools have helped us stay ahead of drug stockouts. The platform is flexible enough for our most complex protocols but still easy for sites to use, which is critical. It’s clear that Sitero values true partnership and understands what operational teams need in a system like this.”
Director, Clinical Supply Chain Operations
GLOBAL PHARMA COMPANY
RTSM Resources
Check out our latest blog posts on clinical supply strategies. More tools and updates coming soon to help you get the most from Mentor RTSM.
In an adaptive oncology trial, the RTSM must do four things that a conventional randomization system is not required to do: enforce configurable cohort and dose assignment rules in real [...]
Biomarker-driven oncology trials enroll slowly because eligibility depends on a chain of steps — tissue procurement, sample shipment, lab processing, result communication, and site decision — each owned by a [...]
Adaptive oncology trials fail operationally, not statistically. Learn what data readiness, RTSM configuration, and DSMB preparation actually require in execution.





