Decades of Clinical Expertise, Engineered for Modern Trials
25+ years of clinical research expertise powering technology-enabled solutions across startup, operations, safety, and compliance. Trusted by 200+ organizations globally.
Core Solutions
Accelerate your clinical trial, drive compliance and safety, and unlock innovation with access to powerful technology and world-class services.
Clinical Services

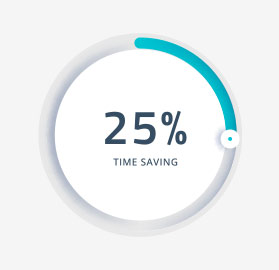
When you partner with Sitero, you can activate your study faster, accelerate trial enrollment, maintain strong relationships with sites and collect higher quality data. Our rapid site activation gets your study running 25% faster than the standard startup time.
Sitero Phase III Study
Traditional Phase III Study
MENTOR eCLINICAL SOFTWARE SOLUTIONS


Latest Blogs, News, & Events
Sitero is excited to be exhibiting at Outsourcing Clinical Trials Southern California 2026 on September 15-16. Learn more:
Sitero is excited to be exhibiting at SCDM US 2026 in Raleigh, NC on September 14-17. Learn more:
Sitero is excited to be exhibiting at Outsourcing Clinical Trials Oncology East 2026 on June 30 to July 1. Learn more:























